Fertigation is a widely used farming practice. It allows growers to save time, resources, and effort by completing two events at a time: fertilization and irrigation. Customization of modern fertigation systems and innovative satellite-based software enable pinpointing of variable rate fertilizer (VRF) applications. The most efficient fertigation systems are built on drip irrigation, which reduces inputs and delivers nutrients to the root zone. The technology is suitable for farm enterprises of any size since there are large and small-scale fertigation systems with manual or fully automated control.
What Is Fertigation?
In fertigation, liquid fertilizers are delivered to plants with irrigation. Compared to traditional fertilization methods, fertigation is more efficient in agriculture. By reducing fertilizer waste and land contamination and increasing crop production, fertigation is a win-win for farmers and the environment.
What does fertigation allow the producer to do? In line with precision farming, it enables agriculture producers to save money by applying fertilizer only where it’s needed. Automated fertigation systems make it simple for farmers to regulate nutrient application rates and schedules.
The most commonly used water-soluble fertilizers for fertigation comprise ammonium nitrate, urea ammonium nitrate, calcium nitrate, ammonium thiosulfate, potassium chloride, potassium sulfate, potassium nitrate, phosphoric acid, sulfuric acid, and so on . Apart from supplying nutrients properly, some fertilizers can perform acidulating functions and boost soil properties.
Advantages Of Fertigation
In general, the targeted application of nutrients through fertigation helps to improve crop yields, quality, and uniformity. Also, fertigation benefits include:
- matching fertilization with crop needs;
- optimizing water consumption;
- saving costs on reduced fertilizer amounts;
- eliminating pollution of the environment by decreasing chemical applications;
- preventing fertilizer leakage due to heavy rainfall or water supply;
- tackling soil erosion;
- encouraging rapid root growth;
- positively affecting soil microbial biomass;
- reducing weed density;
- promoting simplified and automated fertilizing procedures.
The fluid form of nutrients itself is one of the many benefits of fertigation. Because plants can absorb liquid fertilizers so quickly after treatment, they are both easily accessible and effective.
Disadvantages Of Fertigation
While fertigation has numerous advantages in terms of crop growth and nutrient control, it does have some drawbacks that should be taken into account as well:
- For a fertigation system to function properly, expensive components, including pumps, injectors, tanks, and controllers, are typically required.
Maintaining a fertigation system calls for routine upkeep, such as cleaning and inspection. - Fertigation success is constrained by the efficacy of the irrigation system. Crops risk not getting sufficient water and nutrients if the system is poorly constructed or operated.
- Over-fertilization can occur during the process of fertigation if the nutritional mix is imbalanced or the system is not well regulated. This can cause serious harm or even death to the plants.
- Fertigation might become a problem in places where electricity is scarce or intermittent.
Whether or not to use precision fertigation, like most agricultural technologies and automation options, typically boils down to the number of acres of the crop being cultivated. While fertigation technologies might not be cost-effective for smaller farmers, for larger agribusinesses, they almost surely would be. Because of their scalability, fertigation systems are a good option for rapidly growing agricultural enterprises.

How Fertigation System Works
Fertigation technology suggests sprinkling fertilizers into the precision irrigation system from reservoirs with water-soluble fertilizers. Typically, it is done with injectors and a pressure-controlled valve. Most fertigation systems are equipped with sensors to measure pH levels and electric conductivity. This way, necessary fertilizer rates can be determined on fields, and fertigation and irrigation system injectors can be set accordingly.
There are several types of fertigation systems, distinguished by their size and scale, management approach, and application strategy.
Size And Scale Of Applications
Fertigation systems come in many sizes and configurations to accommodate a wide range of applications. Large-scale fertigation systems are used at large enterprises to uniformly improve the soil and thus the yield of their crops. Correspondingly, small-scale fertigation systems are suitable for smaller farms or greenhouses. In these agricultural areas, field blocks are typically between 0.1 and 1 acre in size, and farmers use drip irrigation systems to deliver nutrients to their crops .
Management Approach
Fertigation control systems can be either manually operated or automated. When growing on a small scale, fertilizer solutions are typically mixed by hand. Every fertilizer batch is measured, dosed, modified, and blended manually. By using timers, which are often incorporated into the irrigation system, automated fertigation ensures that every single crop gets the appropriate amount of water and nutrients at precisely the right time in relation to its growth stage. In addition, automated fertigation systems accomplish it without spending the tens of thousands of man-hours needed to manually prepare and administer each nutrient formula.
Process Of Fertilizer Application
The crop, soil type, and farming methodology all play a role when choosing the fertigation fertilizer application approach . Thanks to developments in automation technology, growers now have access to the data and tools they need to implement the optimal approach for their fields. There are four common fertigation methods of fertilizer application:
- Continuous application. From the beginning until the end of the fertigation process, fertilizer is applied at an even rate. The complete dose of fertilizer will be injected no matter how quickly the water drains.
- Three-stage application. Fertilizers are not used at the outset of irrigation. The injection begins once the soil has soaked up enough water and ceases before the irrigation cycle concludes. The fertilizer can then be washed out of the irrigation network for the remainder of the watering cycle.
- Proportional application. There is a direct correlation between the water supply and the rate of injection. In proportional fertigation, one liter of solution per thousand liters of irrigation water, for example, would be considered a standard injection rate. This approach is advantageous since it is easy to implement and can boost fertigation during water-intensive times, which are also the most nutrient-intensive.
- Quantitative application. Each fertigation zone receives a specific dose of nutrient solution based on its individual needs; say, zone A receives 20 liters and zone B receives 40 liters. This approach is amenable to automation and gives you complete command over where the nutrients go.

Fertigation Compatibility With Irrigation Systems
As fertilizers are liquid, their delivery and distribution correlate with wetting patterns. In other words, nutrients will transfer to the areas within water’s reach. Surface, drip, and sprinkler/spray irrigation are all efficient ways to get fertilizer to crops, and each has its own benefits.
Surface Irrigation
Surface irrigation is the most common type of water saturation, applied to 90% of all irrigated lands. However, it might not be a cost-efficient fertigation method as only 30–70% of water reaches the active root zone.
Typically, fertigation systems are not incorporated into surface irrigation since fertilizers are usually supplied via designated canals in established volumes. The equipment includes reservoirs with valves or openings for liquid and solid fertilizers, respectively. It differs by operation complexity (from manual to fully automatic).
Drip Irrigation
Drip irrigation is an example of a pressurized irrigation technique. Pressure-based irrigation, as the name suggests, employs a pressure gradient force to move water and nutrients. A piping and emitter system brings the necessary nutrients to the plant’s root zone in this way. The drip fertigation system is the most efficient option for crop cultivation as it:
- brings nutrients directly to the root zone, which optimizes water and fertilizer use;
- requires less pressure compared to other techniques;
- allows different automation settings.
Typically, drip irrigation wetting patterns are oval or hemispherical, either on the soil surface or at the emitter level under it (depending on if the tape runs on or below the surface). The highest amount of water (and correspondingly, nutrients) will be around the emitter and under it. Horizontal spreading of moisture is conditioned by the soil properties, irrigation rate, and duration, according to plant needs.
EOSDA Crop Monitoring
Access high-resolution satellite images to ensure effective fields management!
Sprinkler And Micro-Sprinkler Irrigation
Fertigation based on sprinkler irrigation is a common type of pressurized nutrient delivery framework. Sprinklers are used to evenly distribute water and nutrients across the plant canopy. Despite its lack of precision compared to drip irrigation, this technique might be effective for fertigating large areas. Strong fertilizers applied through sprinkler systems pose risks for agronomists, including canopy burns and the rusting of metal components.
Micro-sprinkler irrigation is a method of watering and fertilizing plants that makes use of low-pressure sprinklers. If well managed, this technique will provide excellent results for plants of small and medium size.
The decision of which method of fertigation to use will depend on numerous criteria, such as plant size and type, local topography and soil fertility, available budget and other resources, and so on.

What To Consider For Successful Fertigation
As mentioned above, fertigation suggests delivering liquid fertilizers via an irrigation system. Yet merely adding them is not enough.
Agronomists take several fundamental properties into account, including solubility, compatibility, acidity, and salinity (osmotic pressure).
Solubility
First of all, the choice of fertilizers depends on their solubility in water. So, the suitable options are:
- solid ones that can properly dissolve;
- liquid ones that are already dissolved.
Different fertilizer types have different solubility capacities. What is more, the degree and speed of solubility also relate to temperature. So, it matters if nutrients can dissolve at the current temperature in the field or not. Thus, the season should be taken into consideration as well, since the solubility rate will differ in the spring and summer.
Besides, some fertilizers may precipitate out of solution when added in high concentrations to hard water or when the temperature drops, e.g., in a cooler season or on cold nights. This property counts when preparing solutions in advance and storing them. Precipitation is characteristic of monoammonium phosphate, urea phosphate, or phosphoric acid. Ammonium nitrate, potassium nitrate, urea, and ammonium phosphate refer to quick, water-soluble fertilizers.
However, there is another significant aspect to bear in mind. Solutions for fertigation systems can be endothermic or exothermic, for instance, the solution temperature either decreases or increases in the process of dissolution. Generally, most nitrogen-based fertilizers take heat from the water, so the solution temperature drops. Consequently, the preparation process will take more time, and more time will mean a cooler liquid and a reduction in the estimated concentration.
The table below features the correlation of the solubility capacity of some synthetic fertilizer compounds (g/L) with temperature, which is essential in smart fertigation technology .
| Compound | 0°C | 10°C | 20°C | 30°C |
|---|---|---|---|---|
| Ammonium nitrate | 1183 | 1580 | 1950 | 2420 |
| Ammonium sulphate | 706 | 730 | 750 | 780 |
| Calcium nitrate | 1020 | 1240 | 1294 | 1620 |
| Di-ammonium phosphate | 429 | 628 | 692 | 748 |
| Di-potassium phosphate | 1328 | 1488 | 1600 | 1790 |
| Magnesium chloride | 528 | 540 | 546 | 568 |
| Magnesium sulphate | 260 | 308 | 356 | 405 |
| Mono-ammonium phosphate | 227 | 295 | 374 | 464 |
| Mono-potassium phosphate | 142 | 178 | 225 | 274 |
| Potassium chloride | 280 | 310 | 340 | 370 |
| Potassium nitrate | 130 | 210 | 320 | 460 |
| Potassium sulphate | 70 | 90 | 110 | 130 |
| Urea | 680 | 850 | 1060 | 1330 |
Compatibility
When it comes to matching several components for fertigation, it is essential to determine whether they are compatible or not. The basic rules are as follows:
- Prepare separate solutions and store them in separate reservoirs if they may enter an undesirable reaction.
- Do not combine phosphorus or sulfur with calcium.
- Do not add chelates to non-chelates.
- Isolate chelates from acids because they break down at an acidic pH.
- The basic rules for mixing fertilizers are to avoid precipitation and reduction of solubility due to chemical reactivity.
Acidity
Solution acidity causes corrosion, which deteriorates metal reservoirs and fertigation system parts. This parameter is assessed as the pH level, and neither too high nor too low is good. Acid solutions have high corrosivity, while alkaline liquids represent the risk of precipitation. Chloride-based chemicals are also notorious for their corrosive properties.
- Diammonium phosphate would create a higher pH than mono-ammonium phosphate.
- Nitric acid would lower the pH of the solution even at relatively low concentrations.
Besides, agronomists consider the soil reaction to fertigation. In particular, muriate of potash or potassium sulfate gives a neutral reaction. With calcium nitrate or potassium nitrate, it is basic. Ammonium nitrate, urea, ammonium sulfate, monoammonium phosphate, and diammonium phosphate produce an acidic reaction. Phosphoric acid applications cause the strongest soil acidity.
Osmotic Pressure
Typically, irrigation water is to a certain degree saline, and adding salt-containing fertilizers contributes to the salinity even more. Salinity relates to osmotic pressure. Negative osmotic potential complicates water absorption by plant roots, which results in a reduction in yield. Crops suffer from osmotic stress and cannot use moisture even when it is available in the soil because it flows from less saline areas to more saline ones. Plants spend more energy absorbing water and nutrients from fertigation, and if osmotic stress is critical, they die. For this reason, administered fertilizers should produce as low an osmotic pressure as possible.
As a rule, the salinity potential of fertilizers is not measured. Electric conductivity and its relationship to osmotic pressure serve as measures of it. Electric conductivity and pH are computed and then compared. It is specific for each chemical substance. For example, ammonium sulfate would generate a higher osmotic pressure in a solution (per amount of total nutrient applied) than ammonium nitrate.
The following table shows the fertilizer properties: electrical conductivity (EC), pH, and nutrient concentration in 10 mmol/L of fertilizer solutions.
| Compound | Nutrient | Concentration (mg/L) | EC (dS/m) | pH |
|---|---|---|---|---|
| Ammonium nitrate | N | 280 | 0.7 | 5.5 |
| Ammonium sulphate | N | 280 | 1.4 | 4.5 |
| Aqua ammonia | N | 140 | 0.7 | 5.5 |
| Calcium nitrate | N | 280 | 2.0 | 6.9 |
| Di-ammonium phosphate | N P | 280 310 | 0.6 | 7.8 |
| Di-potassium phosphate | P K | 310 780 | 1.9 | 9.2 |
| Magnesium chloride | Mg | 240 | 2.0 | 6.8 |
| Magnesium sulphate | Mg | 240 | 2.2 | 6.9 |
| Mono-ammonium phosphate | N P | 140 310 | 0.4 | 4.7 |
| Mono-potassium phosphate | P K | 310 390 | 0.7 | 4.6 |
| Nitric acid | N | 140 | 0.7 | 2.0 |
| Phosphoric acid | P | 310 | 0.4 | 2.3 |
| Potassium chloride | K | 390 | 0.7 | 7.0 |
| Potassium nitrate | N K | 140 390 | 0.7 | 7.0 |
| Potassium sulphate | K | 780 | 0.2 | 7.0 |
| Urea | N | 280 | 2.7 | 7.0 |
Adsorbility
Another aspect that influences nutrient distribution is their type and ability to adsorb onto soil components. For example, nitrates and sulfates do not adhere to soil particles, while potassium and phosphorus do. In particular, phosphorus binds with calcium or aluminum, and positively charged potassium reacts with negatively charged clay.
Taking into account the aforementioned factors and properly selecting the timing of fertilizer application will allow you to achieve maximum fertigation efficiency.

Fertigation Scheduling
Crops require different nutrient volumes at different plant growth stages. Either too early or too late, applications almost turn into waste due to runoff or volatilization. This particularly refers to nitrates that are not retained in the soil. As for phosphorus, it may leak too, though in many cases, about 50% of this fertilizer is administered before planting.
Fertigation allows agronomists to supply nutrients to crops in the right amount and at the right time, thus proving to be the most efficient method. Increasing the frequency of root-zone fertigation enhances the soil’s capacity to absorb nutrients and stimulates a production increase. This way, fertigation also promotes rapid root growth and boosts root density in the top layers of soil .
Furthermore, smaller fertilizer amounts applied right on time save farmers money and prevent unjustified soil salinization due to saline water or when fertilizers salt out. Soil compaction can also be avoided by using methods like drip irrigation, which do not necessitate the use of heavy machinery to disturb the soil in order to provide nutrients to the plants.
Depending on the crop’s needs within the specified time frame, the nutrient management strategy may call for fertigation on a daily, weekly, or monthly basis. It also makes sense to apply nutrients slightly before the crop needs them to secure successful growth. Typically, the most intensive fertigation period should be during plant growth, with reduced or completely stopped fertilizing at the harvesting stage. By tracking weekly progress, farmers can schedule fertigation events.
Site-Specific Fertigation With EOSDA Crop Monitoring
Modern fertigation systems have a wide array of customization features. This benefit enables farmers to step aside from uniform field treatment because, typically, different field zones have different nutrition needs. Generally, fertilizer rates depend on multiple factors, including :
- type of crop,
- growth stage,
- soil type,
- fertilizer grade,
- solution concentration,
- soil moisture,
- soil temperature,
- osmotic potential,
- fertigation effect on soil microbial biomass (microbial action).
Farmers can identify zones in each field and manually set an appropriate amount of fertilizer for every zone (dividing a field into up to seven vegetation zones). Vegetation maps give actionable insights based on recent satellite data. EOSDA Crop Monitoring shows the field productivity (or lack of productivity) in different zones with different colors. Thus, green highlights the areas with the healthiest vegetation. Red reveals the least healthy crops, summoning immediate attention.
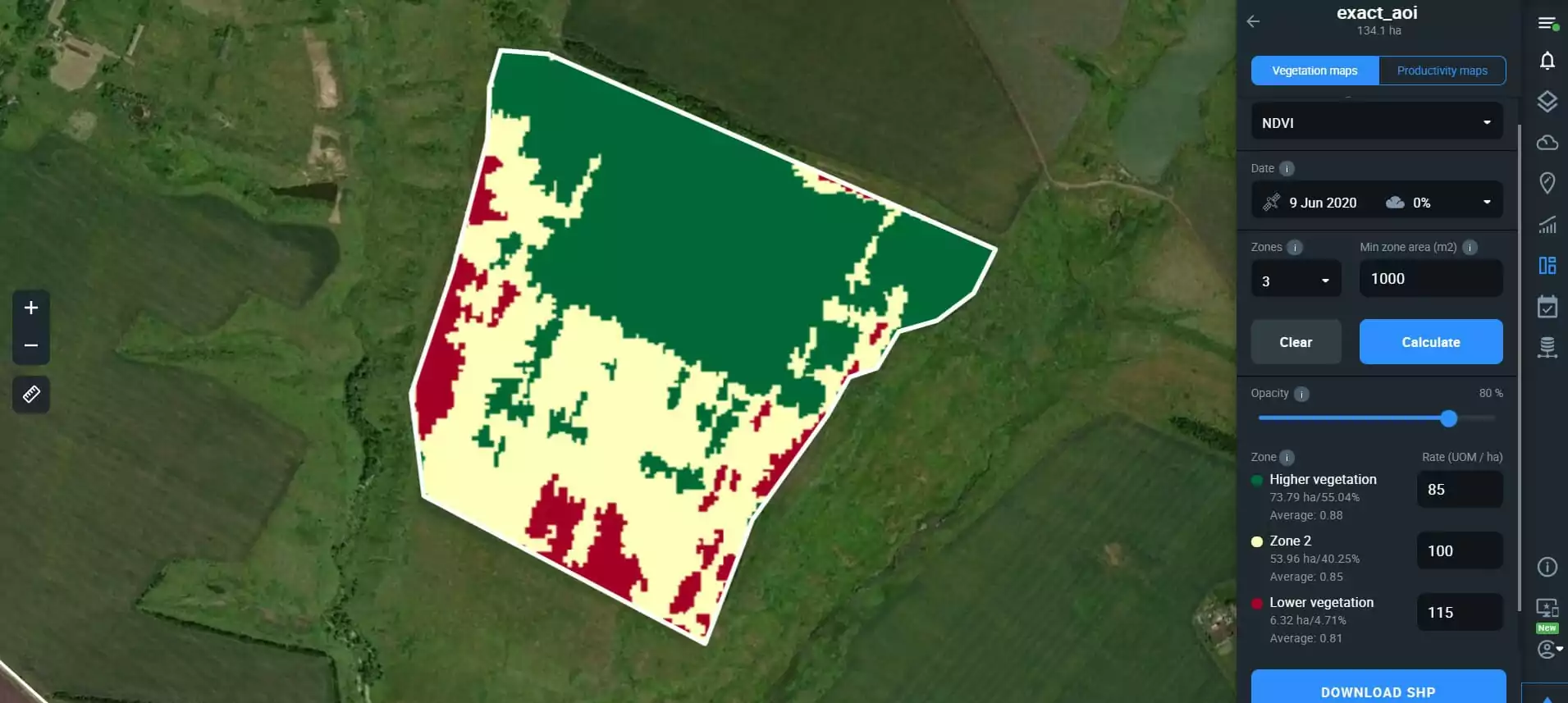
The possible reason for deviance may be a lack of nutrition. So, green areas require a comparatively low fertilizer rate, while red ones require the biggest dose. Furthermore, vegetation and productivity maps facilitate fertilizer input calculations, depending on the field’s needs. Growers enter fertilizer volumes for each zone and get the computed total amount for the field.
Correct fertilizer distribution essentially depends on weather conditions (air temperature, precipitation, wind speed, etc.). In fertigation sprinkler systems, winds may move the mist in the wrong direction, and high concentrations may negatively impact the crops (e.g., burn leaves or fruits). EOSDA Crop Monitoring offers precision weather forecasts for up to fourteen days ahead. Armed with wind speed information, soil fertility specialists can plan applications more efficiently, omitting fertigation waste and unintentional crop leaf and fruit damage.
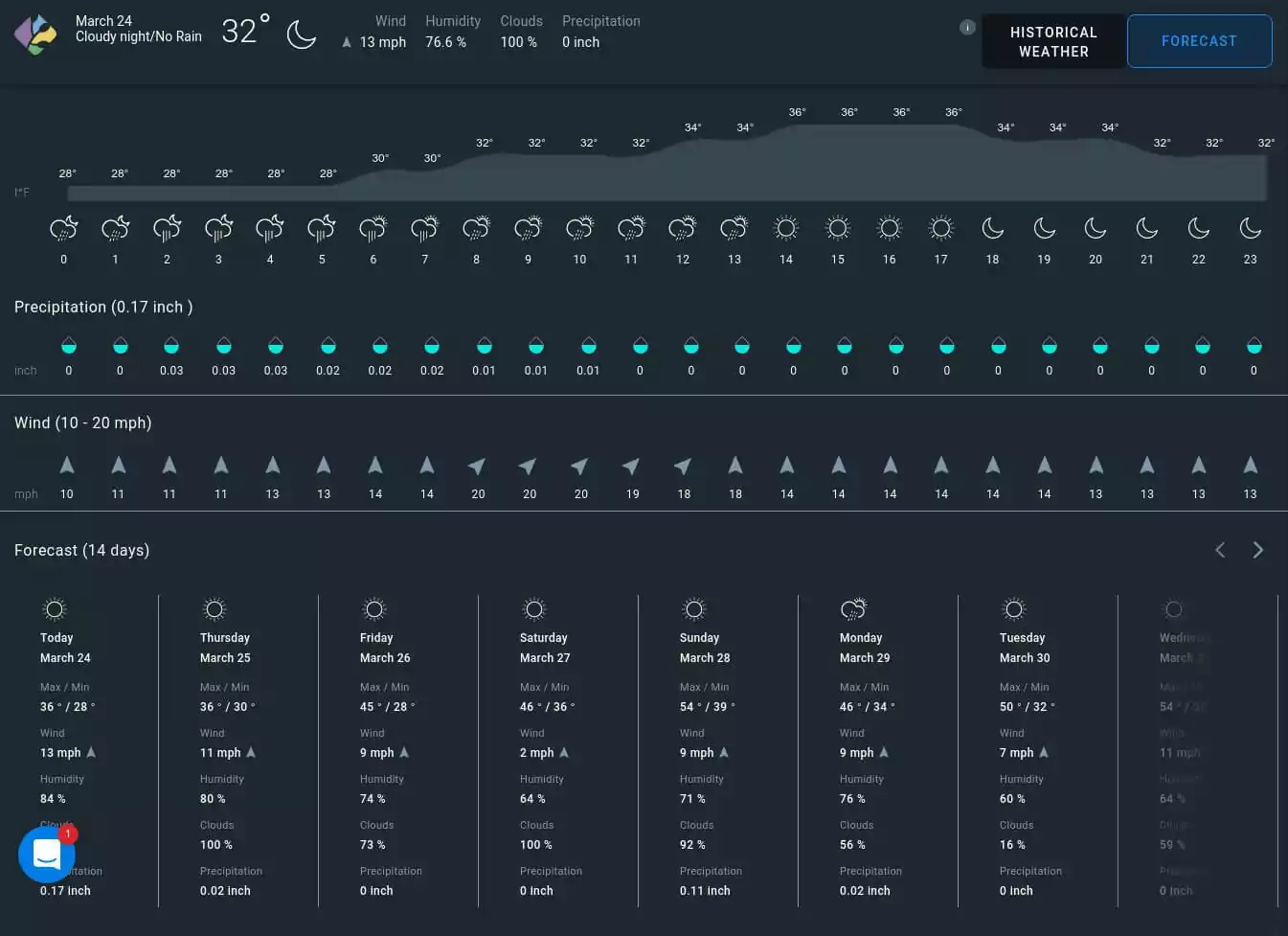
Thus, satellite-retrieved data from the EOSDA Crop Monitoring software makes fertigation more accurate. It is a reliable assistant for every supporter of precision agriculture techniques.
About the author:
Vasyl Cherlinka is a Doctor of Biosciences specializing in pedology (soil science), with 30 years of experience in the field. He attended the engineering college in Ukraine and received his degree in agrochemistry, agronomy and soil science in the Chernivtsi National University. Since 2018, Dr. Cherlinka has been advising EOSDA on problems in soil science, agronomy, and agrochemistry.
More news

Grid Soil Sampling & Its Role In Precision Agriculture
Grid soil sampling is nothing new, but satellites make it easier and cheaper: improve pre-sampling planning, help collect probes efficiently, and combine the results with other soil and crop data.

Yield Monitoring And Mapping In Precision Farming
Why do two neighboring field areas produce such different results? Yield monitoring and mapping help explain why inputs go to waste in some areas and how to manage them more efficiently.

Variable Rate Irrigation Technology And Its Benefits
Variable rate irrigation systems are becoming more affordable and user-friendly. Learn how to get the practical benefits of the technology beyond just water conservation.